
In a the latest research published in Character Chemistry, experts claimed the synthesis of a novel nitride with metallic luster and hexazine rings—the final result of a six-12 months energy in significant-force science.
This is the initially time that a planar N62-, a dianionic hexazine nitride, has been obtained in a laboratory experiment. On top of that, the structure remained reasonably secure at pressures down to 20 GPa.
Nitrogen-wealthy compounds have captivated wide attention because of their wonderful probable as high-electricity density resources (HEDMs) that can shop and release huge amounts of vitality. Having said that, incredibly couple nitrogen compounds have been synthesized so far in comparison with the range predicted theoretically by way of calculation and modeling.
“Lower-order N-N bonds are difficult to hold steady at minimal tension,” claimed Dr. Wang Yu, direct creator of the examine and a researcher at the Hefei Institutes of Bodily Science of the Chinese Academy of Sciences. According to Wang, that is why synthesizing nitrogen compounds in the laboratory is so challenging.
In former research, Wang and her colleagues acquired that molecular diatomic nitrogen can be converted into an atomic stable with a one-bond crystalline cubic gauche (cg-N) framework in a diamond anvil at extreme pressures up to 110 GPa and 2,500 K. The result impressed the synthesis of polynitride elements at higher stress and high temperature, such as the results in their experiments.
Even so, how to manage compound steadiness and restore species underneath useful problems still remains complicated.
In the current review, the researchers made the decision to use alkali metals that have been shown to reduce the force expected for synthesis, as a result improving the stability and power density of possible HEDM compounds.
Researchers experienced also learned that employing linear N3– teams as precursors not only drastically lessens the activation barrier, but also supplies a additional uniform response surroundings.
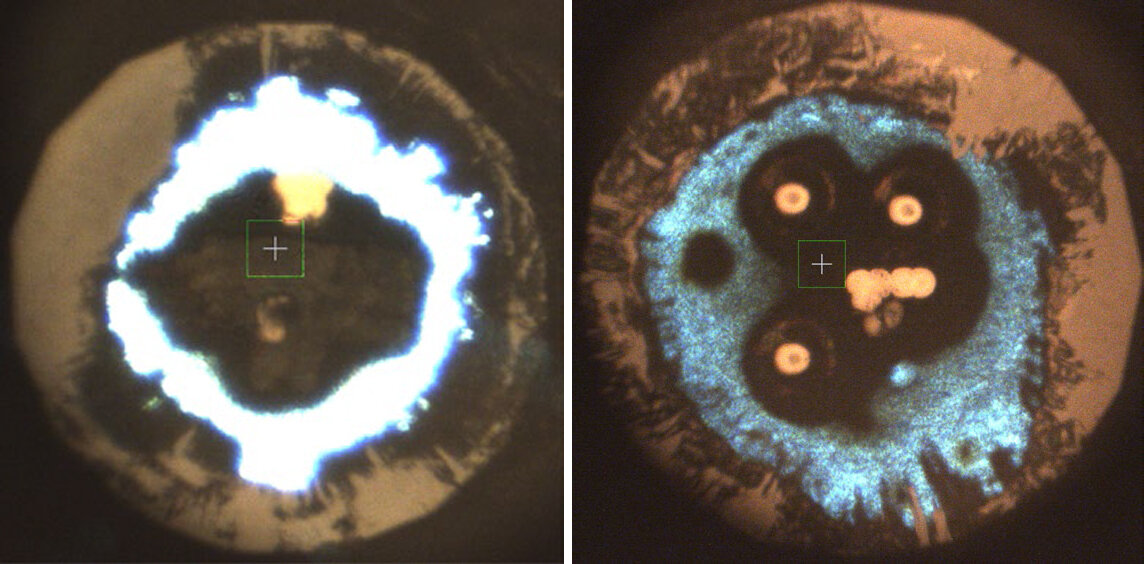
The researchers therefore started their synthesis employing alkali metals and linear N3– azides. They commenced with investigation of potassium azide (KN3), which they put in a diamond anvil chamber applied for X-ray diffraction and Raman experiments.
Via trial and error, the researchers synthesized the N62- plane in the laboratory and stabilized the newly synthesized structure in K2N6. The prosperous synthesis was primarily based on KN3 heated by a laser whilst in a diamond anvil cell at pressures over 45 GPa.
Utilizing X-ray diffraction and Raman detection effects, the scientists, in collaboration with a crew led by Artem Oganov at Skolkovo Institute of Science and Technological know-how, identified that all their success ended up consistent with theoretical predictions by way of very first-ideas calculations.
Wang claimed the researchers ended up “are all really energized” to achieve a nitride with N62 hexazine rings for the 1st time in a lab experiment.
Alexander Goncharov, Stabilization of hexazine rings in potassium polynitride at substantial stress, Character Chemistry (2022). DOI: 10.1038/s41557-022-00925-. www.nature.com/content/s41557-022-00925-
Quotation:
Scientists synthesize novel nitride and stabilize its hexazine rings at significant pressure (2022, April 21)
retrieved 22 April 2022
from https://phys.org/information/2022-04-scientists-nitride-stabilize-hexazine-significant.html
This document is subject matter to copyright. Aside from any reasonable dealing for the function of non-public analyze or study, no
section might be reproduced without having the prepared authorization. The material is provided for information and facts purposes only.





More Stories
Destination Weddings: Your Ultimate Guide to Saying I Do Away
Elopement Videography: Locations, Costs, and Why Couples Elope
TaylorMade Spider GT Rollback, Notchback and Splitback putters